
Newsletter
Sign up and stay in-the-know about The Crowd & The Cloud and the world of citizen science.


My name is Chris Schaffer. I’m an associate professor in the Meinig School of Biomedical Engineering at Cornell University and my current research focus is the role of brain blood flow disruptions in the development of Alzheimer's disease.
Tell us a bit about your work?
We think that if we could improve brain blood flow to Alzheimer’s patients they would already have better cognitive function, and there's a possibility that it could slow progression and delay the onset of the disease. Our goal is to try to understand the mechanisms that lead to this reduced brain blood flow so that we can develop therapeutic strategies to try to combat it.
Alzheimer's disease looks to be caused by a small molecule in the brain called amyloid beta. Amyloid beta is produced by neurons and is usually cleared out of the brain through the vasculature. Individual molecules of amyloid beta don't cause cell injury, death, or any problems, but amyloid beta is a molecule that's very self-sticky. If several of these molecules stick together, they form this big, sticky plaque that binds to all kinds of things in the brain. That causes dysfunction and death in neurons; they don't communicate with each other properly, leading to loss of cognitive function.
Our work is focused on trying to understand how the aggregates of this small molecule, amyloid beta, act to decrease brain blood flow in Alzheimer's disease.
Your lab is doing what hasn’t been seen anywhere else in the world. How is actually looking at those brain blood vessels in living organisms (live mouse brains) helping you better define what is essential to your research?
We study mice that have been genetically engineered to get Alzheimer's disease, so they develop the loss of cognitive function analogous to that found in humans with the disease. In these mice, we remove a small section of their skull and replace it with a thin piece of glass. Then we use microscopes that we developed that allow us to image with micron-scale resolution. A micron is about one one-hundredth the diameter of a human hair.
We can see, with that degree of spatial resolution, down deep into the brain of a living mouse, down a millimeter into the brain of the mouse, which is all the way through the cortex. We can directly visualize all of the blood vessels in the brain. We can see blood cells moving through those blood vessels. We can see the neurons. We can see the inflammatory cells. This allows us to correlate how the aggregates of amyloid beta impact the function of blood vessels in the brain.

One of the reasons that people weren't able to identify the mechanism that leads to the reduced brain blood flow is they just didn't have tools that allowed you to be able to see blood flow at the level of individual capillaries in the brain. Using our tools, we found that mice with Alzheimer's disease had a subset of capillaries with stalled blood flow. It's only a few percent of them, but a few percent of vessels just had no blood flow in them.
In comparison, the normal mice that don't have Alzheimer's disease, basically every capillary in the brain is flowing all the time. A few percent of blood vessels with plugged blood flow doesn't sound like a big deal, but remember, if one blood vessel is plugged, then there's two more downstream that have lost their source of blood flow. By looking at the structure of the vascular network, we predicted that a few percent of the vessels stalled would cause about a 30 percent reduction in brain blood flow; the same degree of blood flow reduction that's found in human patients with Alzheimer's disease.
What's the analysis that you have grad students, postdocs, and undergrads doing to actually track the stalled blood vessels?
These experiments are conducted by taking image stacks, where we image one plane in the brain of the mouse, and then we step down and take another image, and so on. In these images we'll see the same capillary segment in a few successive frames. In vessels that are flowing, we'll see blood cells move from frame to frame, while in a vessel that's stalled the blood cells stay in place.
We worked hard to develop automated image processing routines that could distinguish those flowing from stalled blood vessels, but weren't successful. We were never able to get an accuracy that was high enough to answer the scientific questions we'd like to. But this is something that humans are just very good at. Humans are very good at pattern recognition, at picking up things that are moving versus things that are staying still inside a cylindrical blood vessel like this.

The way that we've handled this analysis is by having large numbers of undergraduates, graduate students, postdocs, myself, and Professor Nozomi Nishimura, my longtime collaborator, just go through and manually score individual capillary segments as “flowing” or “stalled.” At this point, we've manually scored over 100,000 individual capillary segments as flowing or stalled in this study of trying to understand mechanisms that lead to reduced brain blood flow in Alzheimer's disease.
Without being on the frontier of the technology and the engineering, you couldn't be on the frontier of the medical research. Tell us about how cutting edge this technology is.
The instruments we use are truly world class. These are things that we designed and built specifically for these kinds of experiments. In fact, about a third of the effort in my lab is developing novel optics-based tools for biomedical research. About two thirds of the effort using those tools to study the cellular mechanisms that drive neurological diseases. These kinds of innovations in the tools, and the techniques, and the technologies we have, are essential to moving forward scientifically.
The instruments that we have in the lab are not for the faint of heart. They don't operate well every day. They're buggy. They take a lot of maintenance. They take a lot of care. They take a lot of expertise to be able to operate. When they're operating well, and with a user who knows how to use them, we can acquire data that can't be acquired elsewhere in the world.
How long does it take researchers to analyze the data?
Right now, for us to acquire the data that could answer one hypothesis, it would take us about a week. To analyze it, would take almost a year.
Why do you, as scientists doing this research, need to enlist the crowd to help solve the analysis?
Because this data analysis is so painstaking and takes so long, we need citizen scientists to come in and help us with this analysis problem, to help us with this thing that humans are very good at - this pattern recognition problem. We'll be able to acquire data in a week. Crowdsource the analysis over the course of the next couple of weeks. And quickly move on to the next question, and then the next question, and then the next question, so that we gain a handle on the molecular and cellular mechanisms that are causing these capillaries to be plugged. Then we could develop therapies that could reverse it, so that we could study the downstream consequences of getting rid of these plugs and improving brain blood flow.
Tell us about how EyesOn ALZ (formerly known as WeCureALZ) can work towards a cure for Alzheimer’s?
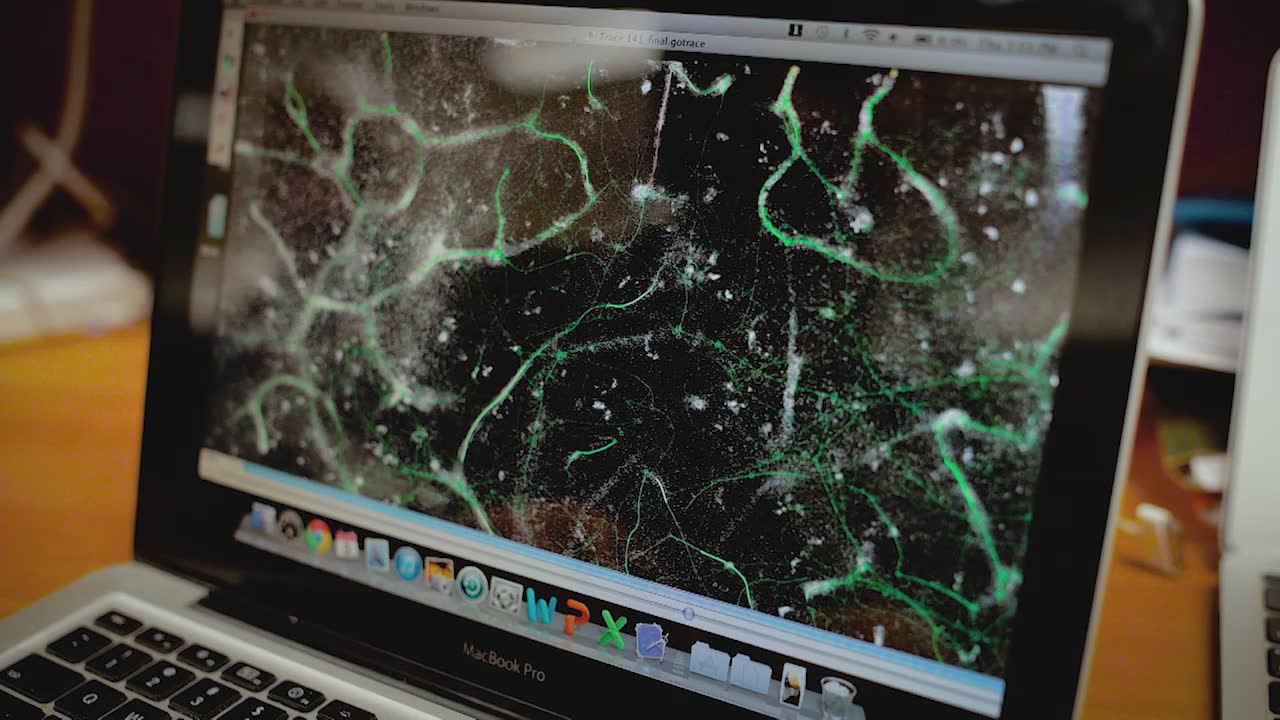
The citizen science project EyesOnALZ aims to take the two really hard parts of this image analysis that we have for this project and to crowdsource those to individuals out in the community to help us with. There's two pieces that we need help with: tracing the vascular network so we know the structure and the topology, and then scoring individual capillary segments as flowing or stalled. One is trying to trace the blood vessels, the path of all of these tiny capillaries through the brain. Once we've traced them, we can then take a segment of an image that has one capillary segment in it. Then we can show that to a user and ask them to assess whether blood cells are moving or not in that capillary segment.
What is it about the disease that you find particularly dehumanizing for those who suffer from it and that you'd like to be part of the solutions to that?
Alzheimer's disease is a terrible disease. Although we've known about it for more than a hundred years, it's really going to be the pandemic of the 21st century. This is a disease that strips away the core of a person's being. It takes away your memories, your ability to conduct yourself in daily life, and your interactions with other people. It's a disease that affects more than just the patients. This is a disease that affects family members and others who care for those who have Alzheimer's disease. Once you have this disease, you cannot take care of yourself. In addition, there is an enormous social cost associated with this disease because it is so expensive to provide care to people who are unable to function or to perform daily functions in life.
You're not an expert on citizen science. Why do you believe that crowdsourcing data analysis is really going to result in high quality data that can inform medical research?
One of the concerns with crowdsourcing a project like this is always the quality of the analysis that we'll get from citizen scientists as compared to what we would be able to do in-house. While I initially had some concerns about this, the results from MalariaSpot are stunning. They've definitely shown that the crowd can produce medical diagnostic information with as good of an accuracy, sensitivity, and specificity as a trained pathologist. That gave me an enormous amount of confidence in the possibility of this project going forward.
The second thing that makes me think that we'll be able to crowdsource this well is that the thing that we're asking people to do here is something that humans are just very good at. It's naturally a sort of pattern recognition problem. We've seen, when we brought new undergraduates or new high school students into the lab, that it doesn't take us very long to get somebody trained where they're able to do this, and this is on our very clunky inhouse software.
By using the Stardust@Home and the EyeWire platform, we'll have great tutorials and great interactive ways to help people develop the skills necessary to be able to do this task.
Why do you believe folks who may not consider themselves citizen science participants will want to jump in given the consequences of this disease on so many families, so many communities, so many individuals?
Everybody wants to cure Alzheimer's disease. Many people make contributions of their money, they volunteer their time for advocacy efforts, and all of that helps. Here's another avenue where people can help, can become involved, and try to work toward developing strategies to ameliorate the devastating consequences of disease, now by contributing your time and your expertise to helping us with the data analysis. We hope this will develop a community of people who we will regard as an extension of our laboratory, who would be involved and kept up to date on making decisions about what kinds of steps to take, and learning about the results. Not just the good results and the things that we're going to move forward in papers, but the experiments that failed, and the ideas that were wrong.
How does your work democratize the understanding of medical research?
One of my hopes is, by having a community of people who are intimately involved in that process with us, that that will help a larger number of people understand how science works. Science works through a very slow, iterative, frustrating process where we're often taking wrong directions, making serendipitous discoveries. One thing I'd like to do is to help people understand that process and help people understand how exciting that process actually is of being off the edge of the map, and really trying to discover something that has never been known before.
